Diclofenac sodium 3% gel
Adverse events should be reported
Reporting forms and information can be found at yellowcard.mhra.gov.uk or downloaded from Google Play or the Apple App Store.
Adverse events should also be reported to JensonR+ on 01271 314 320 or Stirling Anglian Pharmaceuticals on 0345 527 0680.
Information for Healthcare Professionals
Diclofenac sodium 3% gel is indicated for the treatment of actinic keratoses (AKs).
For use in Adults, Diclofenac sodium gel should be applied to the affected skin areas twice daily and smoothed into the skin gently. The amount used depends on the size of the area to be treated.
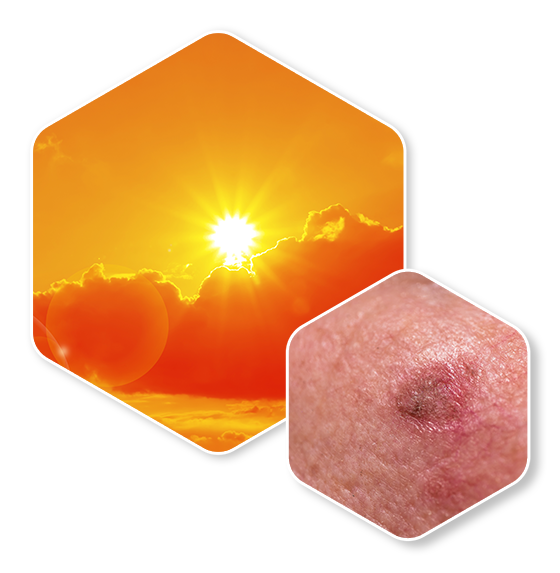

Usually 0.5 g of gel (about the size of a pea) is applied to a 5×5 cm lesion site. The maximum daily amount of 8 grams of product allows simultaneous treatment of up to 200 cm² skin surface area. The usual duration of treatment is 60 to 90 days. Maximum effect has been observed with treatment durations at the upper end of this time range.
Complete healing of the lesion(s) or optimal therapeutic effect may not be seen for up to 30 days after completion of therapy.
AK is a condition not generally seen within the paediatric population and was not studied. Therefore, dosage recommendations and indications for the use of Diclofenac sodium have not been established for use in children and adolescents.
After applying, the hands should be wiped on a paper towel and then washed, unless they are the area to be treated. The paper towel should be disposed of in the residual waste. This helps to significantly reduce diclofenac release into the waste water system (water pollutant).
Diclofenac sodium 3% gel availability
- Legal category: POM
- EU manufactured
- 3 year shelf life (6months after opening)
- NHS list price: £36.38 (50g tube)
- Exclusively stocked and available through Alliance Healthcare, AAH and Phoenix Medical Supplies.
